4.1. Transcriptomics-Proteomics
Pairwise integration and alignment were performed on the multi-omics mouse cerebellum dataset, specifically between the PLATO spatial proteomics and MALDI-MSI spatial metabolomics consecutive slices.
The MAGIC-seq slice captured 1,677 spatial spots at a resolution of 32 μm, profiling 16,116 genes. The PLATO slice provides high-throughput proteomic profiling aligned with the transcriptomic slice, identifying 5,722 protein groups after AI enhancement and quality control.
Preparation
[1]:
import sys
import time
from spatialFuser import *
import scanpy as sc
import pandas as pd
import numpy as np
sys.path.append("..")
/home/whcai/anaconda3/envs/PyG/lib/python3.9/site-packages/louvain/__init__.py:54: UserWarning: pkg_resources is deprecated as an API. See https://setuptools.pypa.io/en/latest/pkg_resources.html. The pkg_resources package is slated for removal as early as 2025-11-30. Refrain from using this package or pin to Setuptools<81.
from pkg_resources import get_distribution, DistributionNotFound
Hyper-Parameters setting
All hyperparameters are stored in the variable args. To display them during initialization, use the function call args = def_training_args(show_detail=True).
For the high-throughput MAGIC-seq slice and PLATO slice, two-layer MCGATE models was employed to learn embeddings.
[2]:
# load args:
print("============================================")
print("= Setting Params =")
slice_1_args = def_training_args()
slice_1_args.hidden = [512, 32]
slice_1_args.epochs = 500
slice_1_args.lr = 3e-3
slice_1_args.K = 4
slice_1_args.heads = 4
slice_1_args.alpha = 0
slice_2_args = def_training_args()
slice_2_args.hidden = [512, 32]
slice_2_args.epochs = 500
slice_2_args.lr = 1e-3
slice_2_args.K = 4
slice_2_args.heads = 4
slice_2_args.alpha = 0
integration_args = def_training_args()
integration_args.hidden = [32, 32]
integration_args.fusion_epoch = 200
integration_args.lr = 3e-3
integration_args.match_step_size = 20
integration_args.tau = 0.1
integration_args.roi_radius = 0.02
integration_args.epsilon = 1
integration_args.m_top_K = 2
integration_args.beta_rec = 50
integration_args.beta_dir = 0.1
integration_args.verbose = False
============================================
= Setting Params =
Load data
SpatialFuser provides a built-in data loading and preprocessing module, SpatialFuserDataLoader. The required inputs include hyperparameters, data_dir (the dataset storage directory), data_tech (either “seq-based” or “image-based”), and files (a list of h5ad files to be loaded).
For spatial omics data, SpatialFuserDataLoader constructs a KNN adjacency graph based on the specified value of K to support graph neural network training.
For seq-based data, spatially variable genes are extracted according to n_svgs to simplify the model.
All AnnData objects are normalized, log-transformed, and subsequently converted into PyG objects for model input.
[3]:
# load data:
print("============================================")
print("= Loading Data =")
slice_1_dataLoader = SpatialFuserDataLoader(slice_1_args,
data_dir='/public8/lilab/student/whcai/Integration/data/cell_fangqing_zhao',
data_tech='seq-based',
files=['Cerebellum-MAGIC-seq_raw.h5ad'])
slice_1_dataLoader.load_adata()
slice_1_dataLoader.pre_processing(n_svgs=3000, k_cutoff=slice_1_args.K, batch_label=[1])
slice_1_dataLoader.generate_minibatch(loader_type='RandomNodeLoader', num_workers=5)
slice_2_dataLoader = SpatialFuserDataLoader(slice_2_args,
data_dir='/public8/lilab/student/whcai/Integration/data/cell_fangqing_zhao',
data_tech='seq-based',
files=['Cerebellum-PLATO_raw.h5ad'])
slice_2_dataLoader.load_adata()
slice_2_dataLoader.pre_processing(n_svgs=3000, k_cutoff=slice_2_args.K, batch_label=[2])
slice_2_dataLoader.generate_minibatch(loader_type='RandomNodeLoader', num_workers=5)
slice_1_dataLoader.adata.obs['Region'] = slice_1_dataLoader.adata.obs['cluster']
slice_2_dataLoader.adata.obs['Region'] = slice_2_dataLoader.adata.obs['cluster']
============================================
= Loading Data =
/home/whcai/anaconda3/envs/PyG/lib/python3.9/site-packages/scanpy/preprocessing/_highly_variable_genes.py:61: UserWarning: `flavor='seurat_v3'` expects raw count data, but non-integers were found.
warnings.warn(
WARNING: adata.X seems to be already log-transformed.
= Calculating spatial graph =
The PyG data u create is qualified
=The graph contains 8385 edges, 1677 cells=
= 5.0000 neighbors per cell on average =
= subgraph Info =
============================================
= Batch 0: 1677 nodes =
= 4.0000 neighbors per cell on average =
batch:[1.], node num:[1677]
============================================
/home/whcai/anaconda3/envs/PyG/lib/python3.9/site-packages/scanpy/preprocessing/_highly_variable_genes.py:61: UserWarning: `flavor='seurat_v3'` expects raw count data, but non-integers were found.
warnings.warn(
WARNING: adata.X seems to be already log-transformed.
/home/whcai/anaconda3/envs/PyG/lib/python3.9/site-packages/scanpy/preprocessing/_normalization.py:196: UserWarning: Some cells have zero counts
warn(UserWarning('Some cells have zero counts'))
= Calculating spatial graph =
The PyG data u create is qualified
=The graph contains 8385 edges, 1677 cells=
= 5.0000 neighbors per cell on average =
= subgraph Info =
============================================
= Batch 0: 1677 nodes =
= 4.0000 neighbors per cell on average =
batch:[2.], node num:[1677]
============================================
/tmp/ipykernel_19184/2482162417.py:20: ImplicitModificationWarning: Trying to modify attribute `.obs` of view, initializing view as actual.
slice_1_dataLoader.adata.obs['Region'] = slice_1_dataLoader.adata.obs['cluster']
/tmp/ipykernel_19184/2482162417.py:21: ImplicitModificationWarning: Trying to modify attribute `.obs` of view, initializing view as actual.
slice_2_dataLoader.adata.obs['Region'] = slice_2_dataLoader.adata.obs['cluster']
Train
The MCGATEs are first pre-trained individually, after which the fusion layer and matching layer are incorporated into the training process.
[4]:
# train
print("============================================")
print("= Begin to Train =")
training_time = time.time()
[adata1, adata2], trainer = train_integration([slice_1_args, slice_2_args, integration_args],
[slice_1_dataLoader, slice_2_dataLoader])
print("= Training Finished! =")
print("Total time elapsed: {:.4f}s".format(time.time() - training_time))
print("============================================")
============================================
= Begin to Train =
/public8/lilab/student/whcai/Integration/model/SpatialFuser/spatialFuser/train.py:137: UserWarning: torch.sparse.SparseTensor(indices, values, shape, *, device=) is deprecated. Please use torch.sparse_coo_tensor(indices, values, shape, dtype=, device=). (Triggered internally at ../torch/csrc/utils/tensor_new.cpp:605.)
self.adata1_adata2atial_adj = torch.sparse.FloatTensor(batch_in_data1.edge_index,
Epoch 500 || adata1_pretrain_loss: 0.128 || adata2_pretrain_loss: 0.053 || : 100%|████| 500/500 [00:29<00:00, 17.16it/s]
Epoch 200 || Fusion_loss : 0.1595 || MSE_loss: 0.1882 || Dir_loss: 0.4295 || : 100%|██| 200/200 [00:12<00:00, 15.88it/s]
= Training Finished! =
Total time elapsed: 43.1083s
============================================
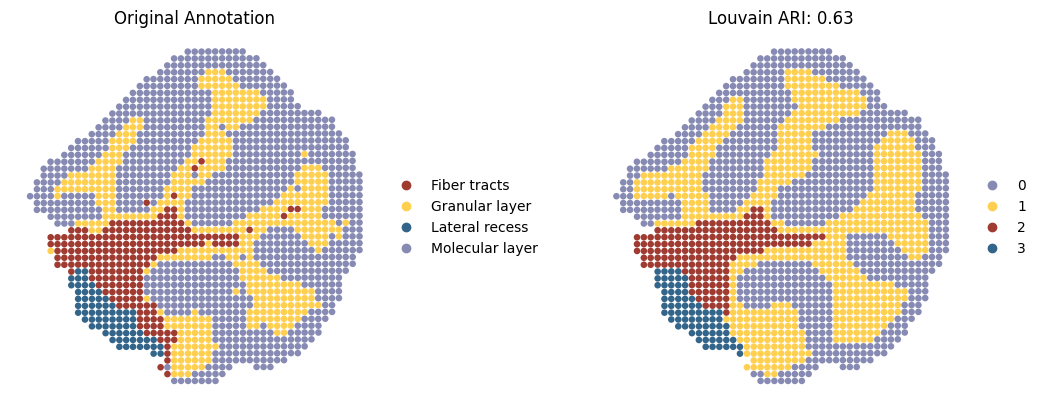
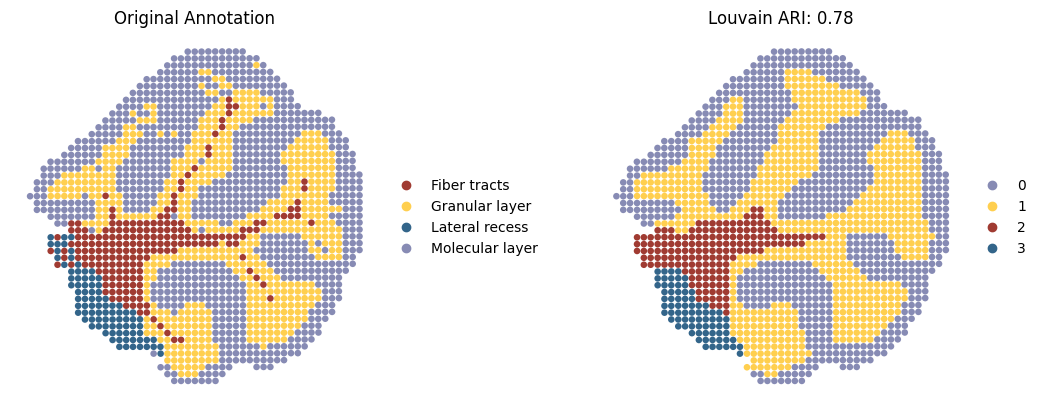
Spatial domain detection after integration
SpatialFuser provides an evaluation module, metrics, for assessing tissue domain detection tasks. It treats the Region column in anndata.obs as the ground truth and, based on the provided embed_label (an array stored in anndata.obsm), automatically computes five metrics (ARI, AMI, Homogeneity, Completeness, and V-Measure) under clustering methods including Leiden, Louvain, and Mclust.
Here, we only present the spatial domains and Umap colored by Louvain.
[5]:
# spatial domain detection
adata1_leiden_result, adata1_louvain_result, adata1_mclust_result = metrics(adata1,
save_loc='_slice1.png',
n_neighbors=10,
resolution=0.1,
spot_size=0.02,
cluster_label='Region',
plot_color=["louvain", ],
mclust_model='EEE',
embed_label='fused_embedding',
vis=False,
save=False)
adata2_leiden_result, adata2_louvain_result, adata2_mclust_result = metrics(adata2,
save_loc='_slice2.png',
n_neighbors=15,
resolution=0.14,
spot_size=0.02,
cluster_label='Region',
plot_color=["louvain"],
mclust_model='EEE',
embed_label='fused_embedding',
vis=False,
save=False)
region_color = {'Fiber tracts': "#a03b32",
'Granular layer': "#ffcf4f",
'Lateral recess': "#33658A",
'Molecular layer': "#878bb4",
'0': "#878bb4",
'1': "#ffcf4f",
'2': "#a03b32",
'3': "#33658A",
1: "#89606a",
2: "#71a2b6",
}
sc.pl.spatial(adata1,
img_key=None,
color=["Region", "louvain"],
spot_size=0.02,
title=['Original Annotation', 'Louvain ARI: {:.2f}'.format(adata1_louvain_result['ARI'][0])],
wspace=0.1,
palette=region_color,
frameon=False,
save='_RNA_Prot_RNA.png'
)
sc.pl.spatial(adata2,
img_key=None,
color=["Region", "louvain"],
spot_size=0.02,
title=['Original Annotation', 'Louvain ARI: {:.2f}'.format(adata2_louvain_result['ARI'][0])],
wspace=0.1,
palette=region_color,
frameon=False,
save='_RNA_Prot_Prot.png'
)
2025-08-25 21:01:18.555406: I tensorflow/core/platform/cpu_feature_guard.cc:193] This TensorFlow binary is optimized with oneAPI Deep Neural Network Library (oneDNN) to use the following CPU instructions in performance-critical operations: AVX2 AVX512F AVX512_VNNI FMA
To enable them in other operations, rebuild TensorFlow with the appropriate compiler flags.
2025-08-25 21:01:18.757985: I tensorflow/core/util/port.cc:104] oneDNN custom operations are on. You may see slightly different numerical results due to floating-point round-off errors from different computation orders. To turn them off, set the environment variable `TF_ENABLE_ONEDNN_OPTS=0`.
2025-08-25 21:01:20.326164: W tensorflow/compiler/xla/stream_executor/platform/default/dso_loader.cc:64] Could not load dynamic library 'libnvinfer.so.7'; dlerror: libnvinfer.so.7: cannot open shared object file: No such file or directory; LD_LIBRARY_PATH: /home/whcai/anaconda3/envs/PyG/lib/python3.9/site-packages/cv2/../../lib64::/usr/local/cuda-12.4/lib64:/public8/lilab/student/whcai/myR/R-4.1.2/lib/
2025-08-25 21:01:20.326483: W tensorflow/compiler/xla/stream_executor/platform/default/dso_loader.cc:64] Could not load dynamic library 'libnvinfer_plugin.so.7'; dlerror: libnvinfer_plugin.so.7: cannot open shared object file: No such file or directory; LD_LIBRARY_PATH: /home/whcai/anaconda3/envs/PyG/lib/python3.9/site-packages/cv2/../../lib64::/usr/local/cuda-12.4/lib64:/public8/lilab/student/whcai/myR/R-4.1.2/lib/
2025-08-25 21:01:20.326510: W tensorflow/compiler/tf2tensorrt/utils/py_utils.cc:38] TF-TRT Warning: Cannot dlopen some TensorRT libraries. If you would like to use Nvidia GPU with TensorRT, please make sure the missing libraries mentioned above are installed properly.
R[write to console]: Package 'mclust' version 6.0.1
Type 'citation("mclust")' for citing this R package in publications.
fitting ...
|======================================================================| 100%
fitting ...
|======================================================================| 100%
WARNING: saving figure to file figures/show_RNA_Prot_RNA.png
WARNING: saving figure to file figures/show_RNA_Prot_Prot.png
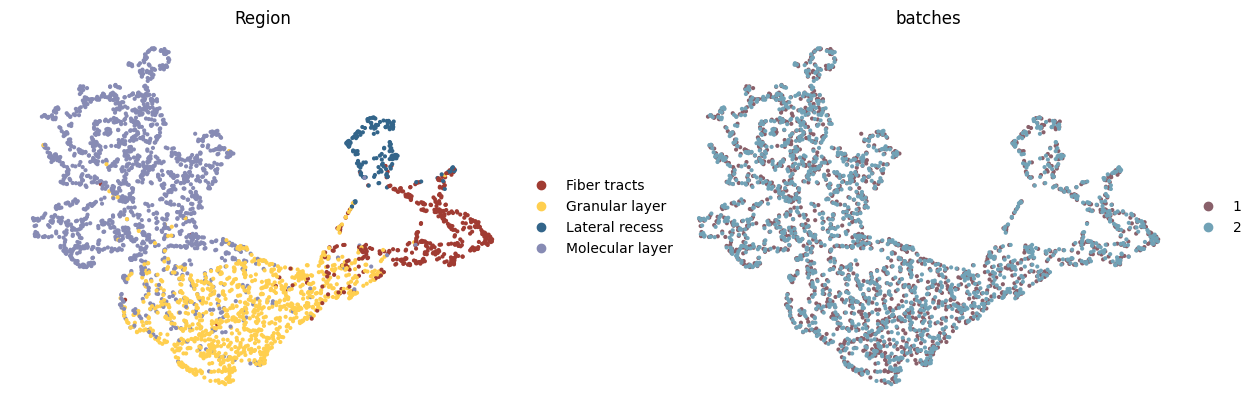
Modality bias correction
UMAP visualizations colored by ground truth annotation (left) and modalities (right), illustrating that the model preserves true biological differences while achieving effective integration.
[6]:
# modality bias correction (integration)
adata = sc.AnnData(X=np.concatenate([adata1.obsm['fused_embedding'], adata2.obsm['fused_embedding']]))
adata.obsm['spatial'] = np.concatenate([adata1.obsm['spatial'], adata2.obsm['spatial']])
adata.obs = pd.concat([adata1.obs, adata2.obs])
adata.obs['batches'] = adata.obs['batches'].astype('category')
sc.pp.neighbors(adata, n_neighbors=15, use_rep='X')
sc.tl.umap(adata, min_dist=0.5, spread=1)
sc.pl.umap(adata,
color=["Region", "batches"],
wspace=0.2,
palette=region_color,
frameon=False,
save='_RNA_Prot_check_batch.png')
WARNING: saving figure to file figures/umap_RNA_Prot_check_batch.png
/home/whcai/anaconda3/envs/PyG/lib/python3.9/site-packages/scanpy/plotting/_tools/scatterplots.py:394: UserWarning: No data for colormapping provided via 'c'. Parameters 'cmap' will be ignored
cax = scatter(
Show alignment
Alignment across slices, colored by region labels (300 alignment pairs shown for clarity)
[7]:
# show alignment
adata1_df = pd.DataFrame({'index': range(adata1.shape[0]),
'x': adata1.obsm['spatial'][:, 0],
'y': adata1.obsm['spatial'][:, 1],
'celltype': adata1.obs['Region']})
adata2_df = pd.DataFrame({'index': range(adata2.shape[0]),
'x': adata2.obsm['spatial'][:, 0],
'y': adata2.obsm['spatial'][:, 1],
'celltype': adata2.obs['Region']})
matching = np.array([trainer.match_in_adata1.data.cpu().numpy(), trainer.match_in_adata2.data.cpu().numpy()])
multi_align = match_3D_multi(adata1_df, adata2_df, matching, meta='celltype',
scale_coordinate=True, subsample_size=300, exchange_xy=False)
multi_align.draw_3D(target='all_type', size=[7, 8], line_width=1, point_size=[0.8, 0.8], line_color='blue',
hide_axis=True, show_error=False, only_show_correct=True, only_show_error=False,
line_alpha=0.5, save=None)
dataset1: 4 cell types; dataset2: 4 cell types;
Total :4 celltypes; Overlap: 4 cell types
Not overlap :[[]]
Subsampled 300 pairs from 1679

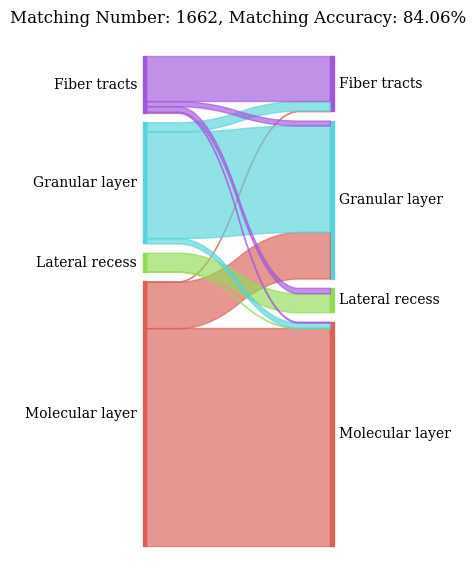
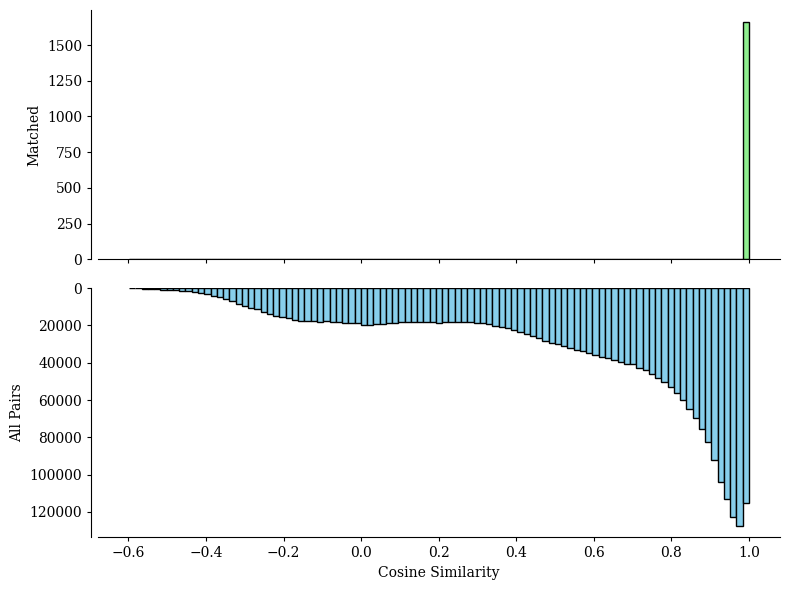
Spots mapping accuracy
The all_matching function returns two plots:
A Sankey plot illustrating spots type correspondence based on alignments from SpatialFuser.
A histogram showing the similarity score distributions from random matching (bottom) and SpatialFuser matching (top) in pairwise alignments.
[8]:
# evaluate alignment
valid_ratio, accuracy = all_matching(adata1, adata2,
0.95,
0.02,
save_loc='./figures/',
file_name='RNA_Prot')
QC threshold:0.95% -> 0.9977
valid matching ratio:99.1% (1662/1677)
spots mapping acc:84.06% (1397/1662)